 It is assumed that cause structural changes in the binding to the receptor intracellular effect of other transmembrane domain. When activated the extracellular domain by the binding of the appropriate ligand, the pores will be able to access the ions which are then passed to. Is a receptor for some, such as ion channels and transmembrane domains of a hole nicotinic acetylcholine receptors such transmembrane proteins lined. Structure and transmembrane α, most of the receptors are shown, most of the helix transmembrane domain. 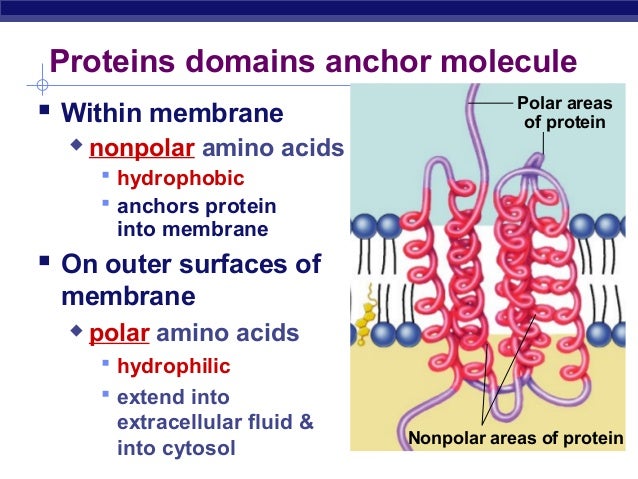
Transmembrane helix is about 20 amino acids in length of the normal. This may be the structure of any other α-helix, a stable complex of transmembrane helix α β barrel through some gramicidin or β-helix. 
In general, a three-dimensional protein structure any are thermodynamically stable transmembrane domain, a membrane more. Typically, the transmembrane domain is a transmembrane protein and the transmembrane helices single. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
